Wprowadzamy na polski rynek innowacyjne produkty zdrowotne.
Wartościowe
relacje biznesowe
Doświadczenie w farmacji
Misja - Innowacja
Polscy pacjenci poszukują innowacyjnych, bezpiecznych i skutecznych wyrobów medycznych, które poprawią ich zdrowie i samopoczucie.
Oferta
Nowy rynek, nowe możliwości.
Wprowadzając produkty na polski rynek zajmujemy się nie tylko marketingiem, ale także kwestiami regulacyjnymi, logistyką, a przede wszystkim budowaniem świadomości wśród lekarzy, farmaceutów i pacjentów.
Służymy doświadczeniem, ugruntowanymi relacjami biznesowymi oraz efektywnymi kanałami sprzedaży, co przekłada się na realną sprzedaż.
Zróbmy razem dobry biznes w Polsce.
Innowacyjne produkty
w asortymencie
Wsparcie marketingowe, regularne promocje, edukacja oraz codzienny kontakt z farmaceutami są naszą wizytówką. Kompetentny zespół REP, świadomy potrzeb i preferencji zarówno aptek sieciowych, jak i butikowych, pomaga w bieżącej sprzedaży produktów.
Budujemy w ten sposób nie tylko wizerunek produktów, ale także relacje z pracownikami aptek i pacjentami.
Dbamy o proces na każdym etapie.
Kosmetyki, suplementy i wyroby medyczne światowych marek.
Poza rozbudowaną siecią sprzedaży hurtowej i bezpośredniej Evalab rozwija alternatywne, własne kanały sprzedaży online.
Marką Evalab Naturals wychodzi naprzeciw obecnym trendom skutecznie torując drogę do odbiorców indywidualnych. Osoby te, szukając skutecznych metod leczenia dolegliwości oraz podnoszenia komfortu życia, sięgają po innowacyjne suplementy i kosmetyki, a także preparaty do aromaterapii.
Znajdź produkty dla siebie.
Na zdrowie.
Sięgnij po kosmetyki i wyroby medyczne
innowacyjnych, jakościowych marek.
Evalab.
Słuszny wybór.
Evalab dostarcza produkty do ponad 10 000 aptek sieciowych i tzw. butikowych w całej Polsce, w tym do aptek przyszpitalnych oraz aptek internetowych. Współpracujemy też z hurtowniami farmaceutycznymi.
Mamy dostęp do największej w Polsce profesjonalnej platformy internetowej dla lekarzy, umożliwiającej zaplanowanie dedykowanych, indywidualnych wizyt handlowych z każdym z nich.
Przedstaw nam swoje produkty. Polscy pacjenci czekają.


Doświadczenie

Wprowadzenie na rynek
marki Osiris
Pod koniec 2020 roku Evalab został wyłącznym dystrybutorem w Polsce szwajcarskiej marki OSIRIS, która wyspecjalizowała się w produkcji i sprzedaży olejków aromatycznych z CBD do użytku zewnętrznego.
Maślan sodu mikro otoczkowany, suplement diety specjalnego przeznaczenia, wprowadzony na rynek w 2009r. promocja u lekarzy specjalistów (Gastrolodzy) w czwartym roku promocji osiągnął 250 000op sprzedaży
Innowacyjne połączenie Paracetamolu z Ibuprofenem lek OTC przeciw bólowy, wprowadzony na rynek od 0 do 100 000op w ciągu 2 lat promocja wyłącznie na poziomie aptek.
Nasze produkty


Przeznaczenie:
• wygodna inhalacja komfort nosa i oczu
• do inhalacji w aerozoloterapii
• do przemywania ran
Skład:
• chlorek sodu 0,9 g,
• woda oczyszczona s.q. 100 ml
Jednodawkowe ampułki z możliwością zamknięcia w celu późniejszego użycia. Higieniczne i łatwe w użyciu.



Żel do masażu dziąseł w tubce 25 ml, przeznaczony dla niemowląt cierpiących z powodu ząbkowania. Skutecznie łagodzi dolegliwości towarzyszące ząbkowaniu, zapewniając spokój dziecku i rodzicom. Bez efektów ubocznych, nie uczula.
Skład:
• Rumianek (zmiękcza i łagodzi)
• Korzeń waleriany (odpręża)
• Propolis (uspakaja i łagodzi)
Bez cukru, bez alkoholu.
Nie zawiera lydocainy/benzokainy.
• Produkowany w reżimie farmaceutycznym
• Bogaty w wapń, dla prawidłowego wzrostu zębów

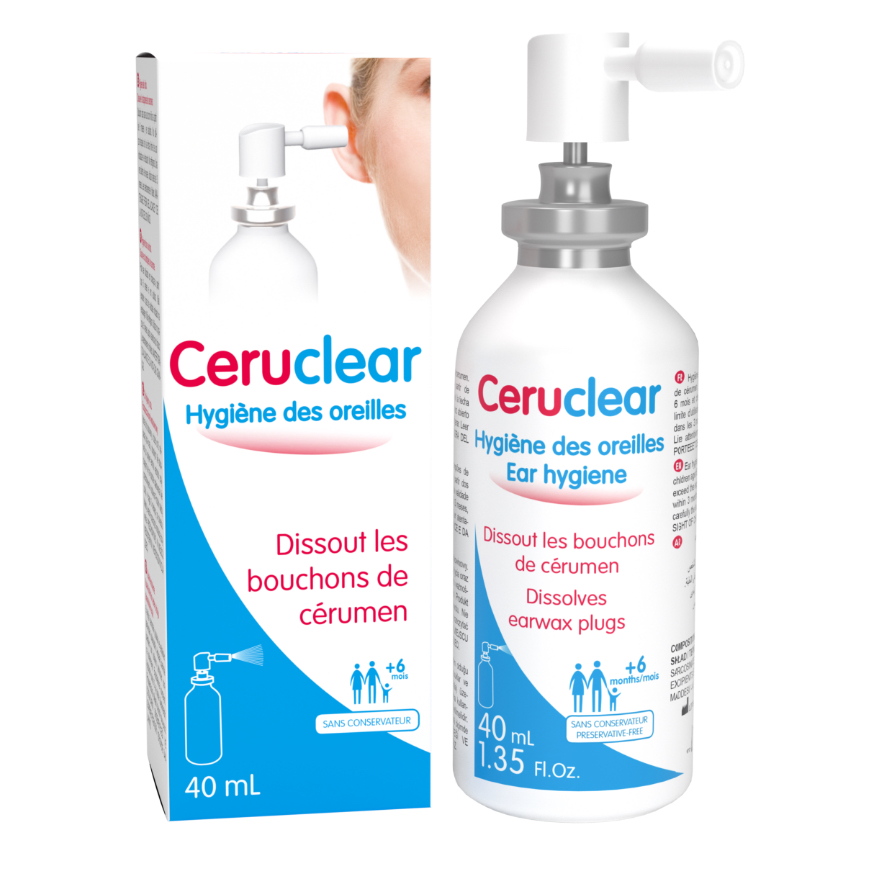

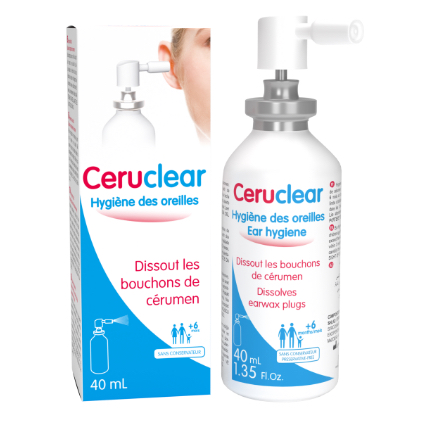
Produkt o podwójnym działaniu: SKUTECZNIE ROZPUSZCZA WOSKOWINĘ ZAPOBIEGA ZBIERANIU WOSKOWINY (do codziennej higieny ucha)
• Łatwy w użyciu, nie podrażnia
• Dla dorosłych i dzieci (od 6 miesiąca)
• PH Ceruclear = PH ucha
• Bez konserwant w i sztucznych barwników



• Oczyszcza i nawilża błonę śluzową nosa
• Zapobiega infekcjom nosa i gardła
• Zapewnia błonie śluzowej nosa warstwę ochronną przed zarazkami, kurzem etc.
• Naturalnie bogata w sole mineralne i pierwiastki śladowe
• Niezbędna dla niemowląt – nie potrafią wydmuchać nosa
• Do stosowania od urodzenia
• Bez konserwantów
• 100% bezpieczeństwa i pewności



Oczyszczenie i udrożnienie dzięki efektowi osmozy
• Udrażnia zatkany nos (przy przeziębieniach, katarze alergicznym,
chorych zatokach)
• Optymalne działanie naturalnych składników przy infekcjach i alergiach
• Zwalcza infekcje i zapobiega nowym
• Zapewnia prawidłowe oczyszczenie jamy nosowej
• SPRAY 100 ml 2.2%: dla lepszej tolerancji
• Do stosowania od urodzenia
• Bez konserwantów
• 100% bezpieczeństwa i pewności



 EN
EN